
Polymerization of monomers into polymers occurs by dehydration reactions, chemical reactions that link two subunits together via a covalent bond while extracting an -OH and an H to create a molecule of water: H2O. One recent study concluded that cells are composed of 68 distinct organic molecules (Marth 2008) that are assembled into 3 biological polymers plus lipid structures (membranes). Small organic molecules are covalently linked (polymerized) to form the 3 types of large biological macromolecules (polymers) lipid membranes self-assemble. Students should know how cells make these macromolecules, and their basic structures and functions.ģ. The 3 types of macromolecules (very large molecules) are polysaccharides, nucleic acids (DNA and RNA), and proteins. 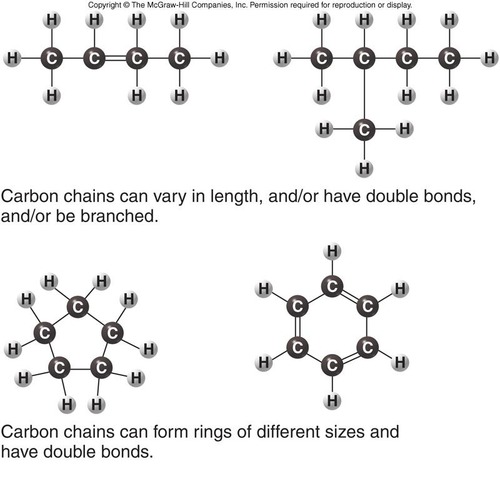
The biomass of a cell (the organic contents, excluding water and inorganic salts) is composed of 3 types of macromolecules plus lipids. The reduced atom has gained a majority share of the electrons that form the covalent bond, and the oxidized atom has only a minority share.Ģ. Recall that a covalent bond is formed when two atoms share a pair of electrons. Conversely, carbon is oxidized when it forms a covalent bond with an atom with greater electronegativity, such as oxygen. Briefly, atoms such as carbon or oxygen are said to be reduced if they form covalent bonds with an atom with lesser electronegativity, such as hydrogen. For a review of oxidation-reduction ( redox) reactions from a biology point of view, see this Khan Academy video. Synthesis of organic carbon molecules from inorganic CO2 requires energy and chemical reducing power, as the carbon atoms in organic molecules are in reduced form. Organic molecules can arise naturally from abiotic synthesis (see Miller-Urey expt), but in the biosphere, most organic molecules are synthesized by living organisms. (An exception to the above rules is urea, where carbon has bonds to 2 amino nitrogens and a double bond with oxygen – but we won’t ask you to remember this exception focus on the general rules above.).Carbon dioxide (CO2 O=C=O) is an inorganic form of carbon because the carbon atom has bonds only to oxygen atoms, and is therefore completely oxidized. In chemistry parlance, the carbon atom in organic molecules must be reduced and not be fully oxidized (has covalent bonds only to oxygen atoms). Organic molecules have at least one covalent bond between C and H or between C and C.Organic molecules must have C and H, and may have O, N, P, S (a handy mnemonic is CHNOPS for carbon, hydrogen, nitrogen, oxygen, phosphorus and sulfur).So how do we recognize organic molecules? Organic compounds are all built from carbon atoms, but not all molecules containing carbon are organic. Up to the early 19th century, scientists thought only living organisms could make organic compounds. One of the distinguishing features of life is that cells are made of organic compounds and large molecules constructed from simple organic compounds. All living organisms are made of organic molecules. You can also read a fuller explanation in the OpenStax Biology textbook (free): 2.1 Atoms, Isotopes, Ions and Molecules: the Building Blocksġ. If you are unfamiliar with them, you should review our web page: Chemical context for Biology. Relate how changes in subunits affect the structure and function of macromolecules (particularly proteins).īefore we begin, we assume that you know these basic chemical concepts:. 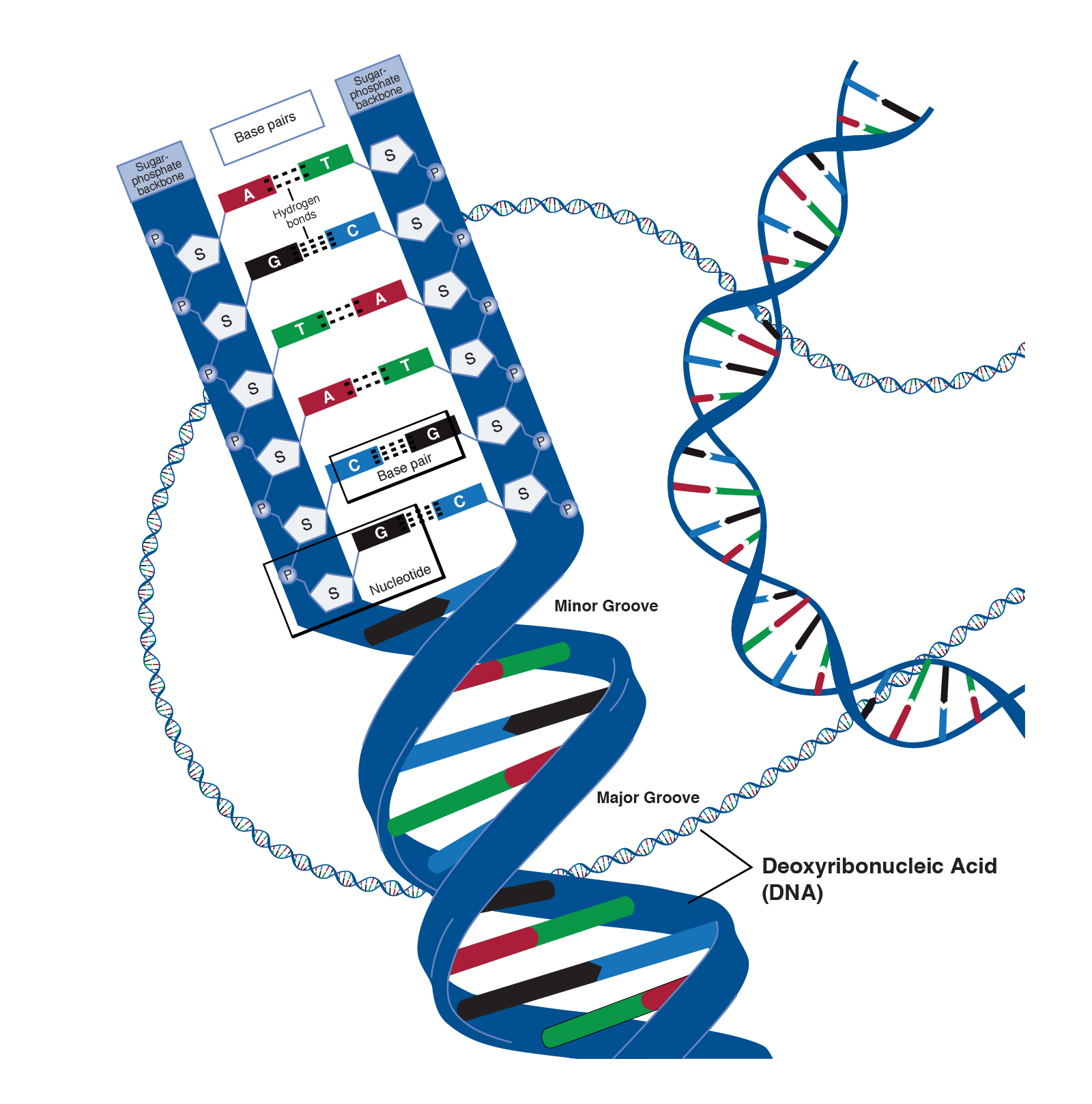
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
